Humanized mice refer to mice stably reconstituted or engrafted with human tissues/cells, such as human immune and liver cells. Humanized mice with a reconstituted human immune system are constructed by engrafting immunodeficient mice with human hematopoietic stem cells (HSCs), which then give rise to all human blood lineage cells throughout the lifetime of the mice.
We have made significant contributions to the development of the humanized mouse technology by i) expanding HSCs in vitro for large scale construction of humanized mice, ii) expressing human cytokines in recipient mice to enhance the reconstitution and function of human immune cells, including myeloid lineage cells, and iii) generating humanized mice with human liver cells and matching human immune system.
The stable reconstitution of human cells in mice provides a unique opportunity to model diseases mediated by human cells in the presence of autologous human immune system. By expressing proto-oncogenes Bcl-2 and Myc in human B-lineage cells, we have constructed double-hit lymphoma in humanized mice that exhibits characteristic histopathological features of the human tumor, including infiltration into the central nervous system. We have evaluated the efficacy of anti-CD52 antibody (alentuzumab) for treating double-hit lymphoma and identified a potent synergy between alentuzumab and cyclophosphamide in eliminating bone marrow residual tumor cells. We have also developed humanized mouse models of infectious diseases for various pathogens that infect only human cells, including dengue virus, Plasmodium falciparum, and Epstein Barr Virus (EBV).
Ongoing projects:
- Molecular mechanisms underlying the progression of human hematologic cancers
- Mechanisms of cancer immunotherapies in mice with human cancer and autologous human immune system
- Molecular and cellular mechanisms of immune responses to dengue virus and malaria parasite
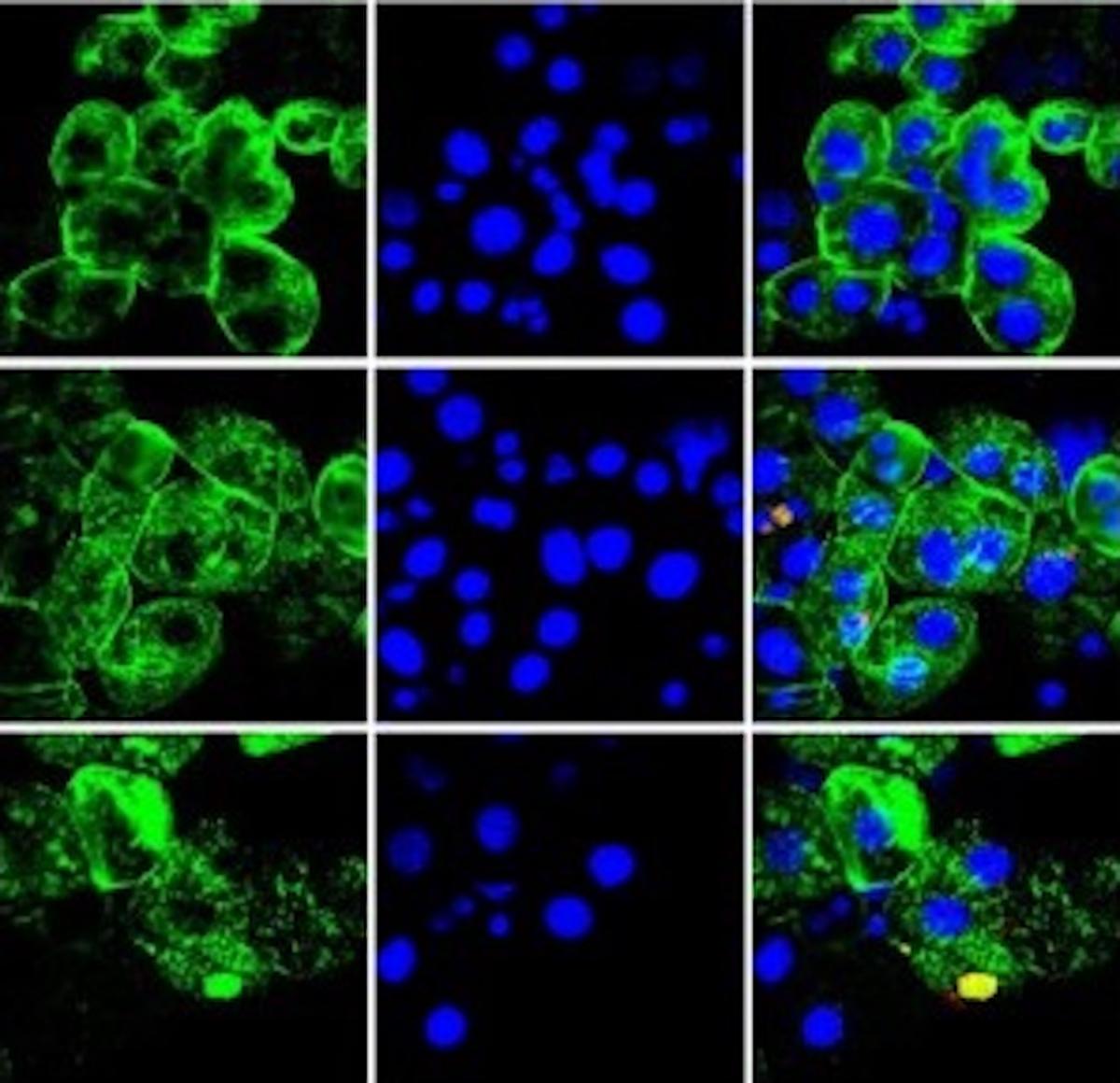
Staining of Cytokeratin 18 (green) and DAPI (blue), markers of cell death, in humanized mice liver cells to investigate the affect of HCV infection (Keng et al. 2016).
Join the team!
We are actively recruiting individuals, including lab technicians, postdoctoral scholars, and graduate students.
Please email Professor Chen (jchen@mit.edu) with your interest and CV/résumé.

